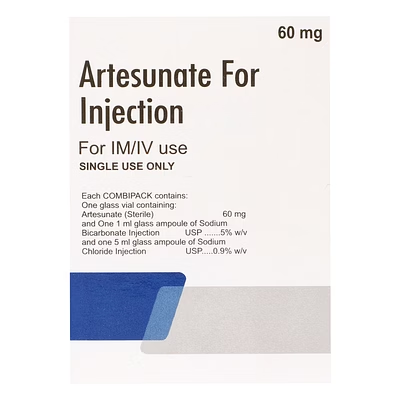
Combipack of Artesunate for Injection 60mg
Artesunate Injection
Artesunate Injection is a biocompatible antimalarial injectable formulation delivering 60mg per pack, designed for rapid parasiticidal action in clinical and hospital environments.
The 60mg specification ensures precise therapeutic dosing for institutional and emergency medical use.
Key Features:
- Rapid systemic antimalarial effect
- Stable under ambient and refrigerated storage
- Low-viscosity solution for smooth administration
Attributes:
- 60mg per pack for predictable therapeutic outcome
- Injectable solution with uniform solubility
- Compliance-ready for bulk hospital and institutional procurement
Ondansetron Injection 8mg/4ml
Ondansetron Injection
Ondansetron Injection is a biocompatible parenteral formulation designed for antiemetic therapy, providing precise 8mg/4ml dosing for rapid onset of action in clinical settings.
The 4ml standardized volume ensures repeatable dosing and consistent therapeutic effect.
Key Features:
- Optimized aqueous injectable for rapid systemic absorption
- 8mg active concentration per 4ml unit
- Maintains stability under ambient and refrigerated storage
- Minimized injection site irritation via controlled pH
Attributes:
- 8mg/4ml for predictable therapeutic efficacy
- Injectable formulation with controlled viscosity
- Regulatory-compliant for professional institutional use
Paraldehyde Injection B.P. 5ml
Paraldehyde Injection B.P.
Paraldehyde Injection B.P. is a biocompatible injectable formulation for rapid central nervous system depressant therapy, delivering 5ml dosage per vial for controlled clinical use.
The 5ml specification ensures precise administration and predictable therapeutic effect for emergency and institutional applications.
Key Features:
- Rapid CNS depressant action with controlled dosing
- Maintains chemical and physical stability under ambient storage
- Standardized solution for reliable therapeutic outcomes
Attributes:
- 5ml per vial for precise dosing
- Injectable formulation with uniform flow characteristics
- Compliance-ready for bulk, hospital, and institutional use
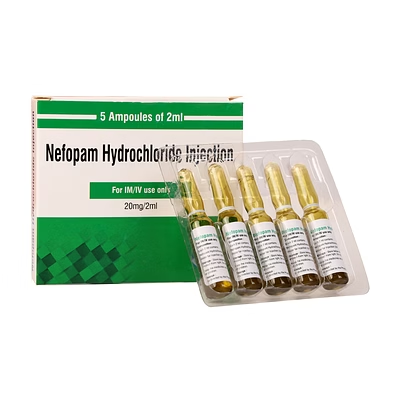
Nefopam Hydrochloride Injection 20mg/2ml
Nefopam Hydrochloride Injection
Nefopam Hydrochloride Injection is a biocompatible parenteral analgesic formulation delivering 20mg per 2ml for rapid pain management in clinical and hospital settings.
The 20mg/2ml specification ensures precise dosing and predictable therapeutic outcomes in institutional applications.
Key Features:
- Rapid systemic analgesic action with controlled dosing
- Stable injectable formulation under ambient and refrigerated storage
- Minimal tissue irritation due to controlled osmolarity
Attributes:
- 20mg per 2ml vial for consistent analgesic effect
- Injectable solution with uniform flow and solubility
- Compliance-ready for bulk, hospital, and institutional use
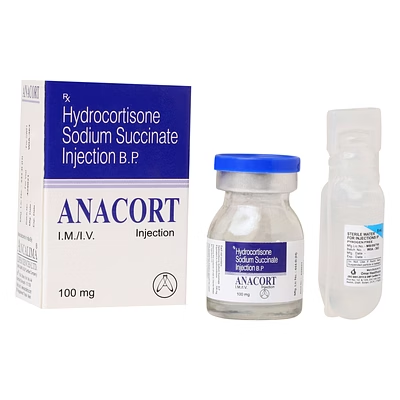
Hydrocortisone Sodium Succinate Injection 100mg
Hydrocortisone Sodium Succinate Injection
Hydrocortisone Sodium Succinate Injection is a biocompatible injectable formulation designed for rapid systemic corticosteroid delivery, providing precise 100mg dosing per vial for clinical applications.
The 100mg specification ensures standardized anti-inflammatory and immunosuppressive therapy, allowing predictable pharmacokinetics in hospital and institutional use.
Key Features:
- Optimized solubility for immediate systemic absorption
- Maintains chemical and physical stability under storage conditions
- Controlled pH and osmolarity for minimal injection site irritation
- Single-use vial for dosage accuracy and safety
Attributes:
- 100mg dosage ensuring consistent therapeutic effect
- Injectable formulation with uniform flow and stability
- Compliance-ready for bulk, hospital, and institutional use
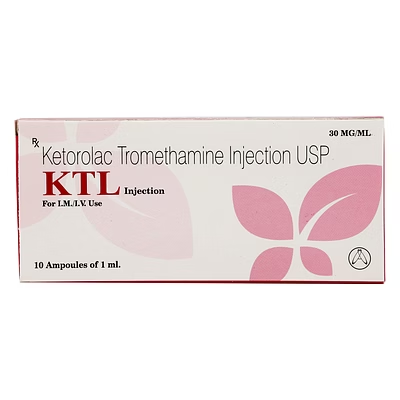
Ketorolac Tromethamine Injection 30mg/1ml
Ketorolac Tromethamine Injection
Ketorolac Tromethamine Injection is a biocompatible injectable NSAID formulation delivering 30mg per 1ml vial, designed for rapid analgesic and anti-inflammatory effect in controlled clinical environments.
The 30mg/1ml specification ensures standardized dosing for predictable pain management in hospital and institutional use.
Key Features:
- Rapid onset analgesic effect with standardized dosing
- Stable chemical composition under ambient and refrigerated conditions
- Controlled pH and osmolarity for injection safety
Attributes:
- 30mg per 1ml vial for predictable therapeutic outcomes
- Injectable formulation with uniform solubility and flow
- Suitable for bulk hospital and institutional procurement
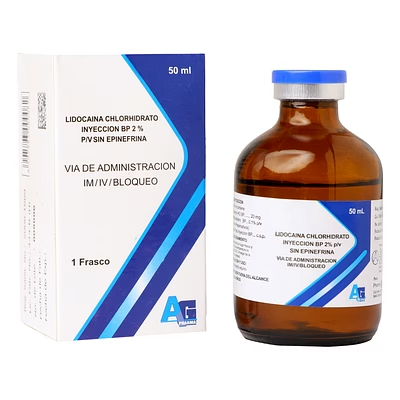
Lidocaine Injection BP 2% w/v
Lidocaine Injection BP
Lidocaine Injection BP is a biocompatible injectable local anesthetic delivering 2% w/v solution, designed for rapid onset analgesia and procedural anesthesia in clinical environments.
The 2% w/v specification ensures standardized anesthetic effect, suitable for surgical, diagnostic, and institutional applications.
Key Features:
- Rapid onset of local anesthesia
- Stable formulation under ambient and refrigerated storage
- Optimized pH and osmolarity for minimal tissue irritation
Attributes:
- 2% w/v concentration for predictable anesthetic effect
- Injectable solution with uniform flow and solubility
- Compliance-ready for bulk hospital and institutional use
Combipack of Artesunate for Injection 120mg
Artesunate Injection
Artesunate Injection is a biocompatible antimalarial injectable formulation delivering 120mg per pack, designed for rapid parasiticidal action in clinical and hospital settings.
The 120mg specification ensures precise therapeutic dosing, suitable for institutional and emergency use.
Key Features:
- Rapid systemic antimalarial activity
- Stable formulation under ambient and refrigerated conditions
- Low-viscosity solution for smooth injection
Attributes:
- 120mg per pack for predictable therapeutic effect
- Injectable formulation with consistent solubility
- Compliance-ready for bulk hospital and institutional procurement
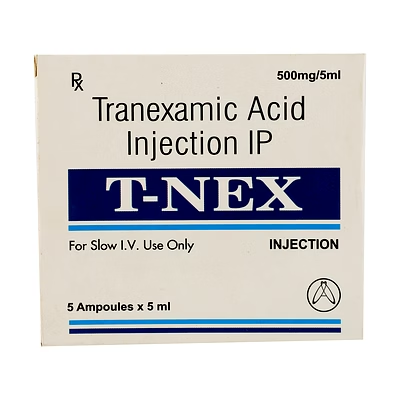
Tranexamic Acid Injection BP (100mg/ml)
Tranexamic Acid Injection BP
Tranexamic Acid Injection BP is a biocompatible injectable formulation designed to inhibit fibrinolysis and stabilize clot formation. The 100mg/ml concentration ensures precise dosing for clinical hemostatic interventions.
This standardized dosage supports rapid hemostatic control, making it suitable for surgical, trauma, and institutional applications.
Key Features:
- High solubility and stability in isotonic solution
- Optimized for rapid systemic fibrinolytic inhibition
- Minimizes injection site irritation due to controlled pH
- Single-use vial ensures dosage accuracy
Attributes:
- 100mg/ml per vial for predictable therapeutic effect
- Injectable formulation with uniform flow
- Suitable for bulk hospital and institutional procurement
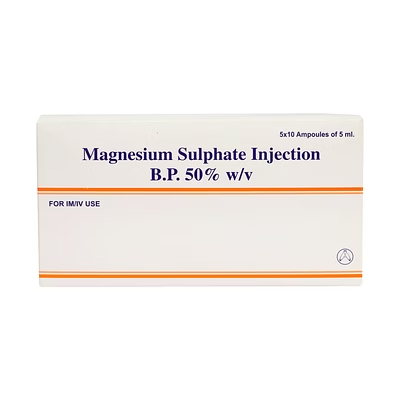
Magnesium Sulphate Injection BP 50% w/v 5ml
Magnesium Sulphate Injection BP
Magnesium Sulphate Injection BP is a biocompatible injectable electrolyte solution delivering 50% w/v in 5ml vials, designed for rapid correction of magnesium deficiency and seizure prophylaxis.
The 50% w/v, 5ml specification allows for precise therapeutic dosing in institutional and hospital settings.
Key Features:
- High-purity magnesium sulphate for rapid systemic effect
- Optimized isotonicity and pH for injection safety
- Stable under ambient and refrigerated conditions
Attributes:
- 50% w/v in 5ml per vial for accurate dosing
- Low-viscosity injectable solution for smooth administration
- Bulk-sourcing ready for hospitals and institutional procurement
Combipack of Pantoprazole for Injection 40mg
Combipack of Pantoprazole for Injection 40mg - Injectable Formulation
Pantoprazole injectable combipack contains 40mg per unit in sterile injectable form. Suitable for professional clinical use and bulk procurement.
Supports distributors, OEMs, and export buyers requiring standardized injectable supply with secure handling.
Key Features
- Sterile injectable combipack
- 40mg per unit
- Bulk and institutional supply ready
Product Attributes & Specifications
- Formulation: Sterile injectable solution
- Composition: Pantoprazole
- Packaging: Combipack format
Magnesium Sulphate Injection BP 50% w/v 10ml
Magnesium Sulphate Injection BP 50% w/v 10ml - Injectable Formulation
Magnesium Sulphate Injection BP provides a sterile 50% w/v solution in 10ml vials. Designed for professional clinical and bulk procurement use.
Suitable for distributors, institutional buyers, OEMs, and export supply requiring standardized injectable formats.
Key Features
- Sterile 50% w/v injectable solution
- 10ml vial per unit
- Bulk and institutional supply ready
Product Attributes & Specifications
- Composition: Magnesium Sulphate BP
- Formulation: Injectable solution
- Packaging: 10ml vial
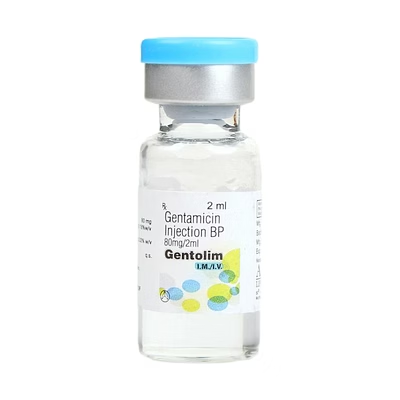
Gentamicin Injection B.P. 2ml (80mg/2ml)
Gentamicin Injection B.P. 2ml (80mg/2ml) - Injectable Formulation
Gentamicin Injection B.P. provides 80mg per 2ml vial in a sterile injectable formulation. Designed for professional clinical settings and bulk procurement.
Suitable for distributors, institutional buyers, OEMs, and export supply chains requiring standardized handling and professional-grade injectable products.
Key Features
- Sterile 2ml injectable solution
- Contains 80mg Gentamicin per vial
- Prepared for bulk and institutional supply
Product Attributes & Specifications
- Formulation: Injectable solution
- Intended for clinical use
- Packaging: single 2ml vial
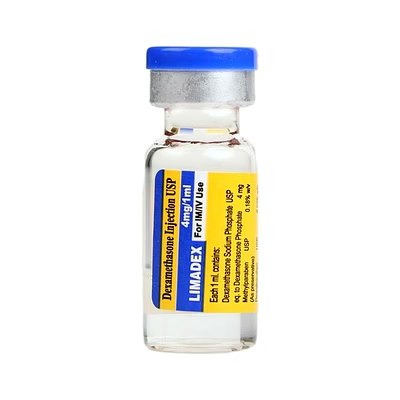
Dexamethasone Injection USP (4mg/1ml) 1ml
Dexamethasone Injection USP (4mg/1ml) 1ml - Injectable Formulation
Dexamethasone Injection USP provides 4mg per 1ml vial in sterile injectable form. Prepared for professional clinical and bulk procurement usage.
Supports distributors, institutional buyers, OEMs, and export buyers needing standardized handling and professional injectable formats.
Key Features
- Sterile 1ml injectable solution
- 4mg per unit dosage
- Bulk and institutional supply ready
Product Attributes & Specifications
- Formulation: USP sterile injectable solution
- Composition: Dexamethasone
- Packaging: 1ml vial
Combipack of Omeprazole Sodium for Injection 40mg
Combipack of Omeprazole Sodium for Injection 40mg - Injectable Formulation
This combipack provides sterile 40mg Omeprazole Sodium per unit in injectable form. Prepared for bulk procurement and professional clinical usage.
Designed to support distributors, OEMs, and export buyers requiring efficient handling and standardized packaging for institutional supply.
Key Features
- Sterile injectable combipack
- 40mg per unit dosage
- Bulk and institutional-ready format
Product Attributes & Specifications
- Formulation: Injectable solution
- Composition: Omeprazole Sodium
- Packaging: Combipack format
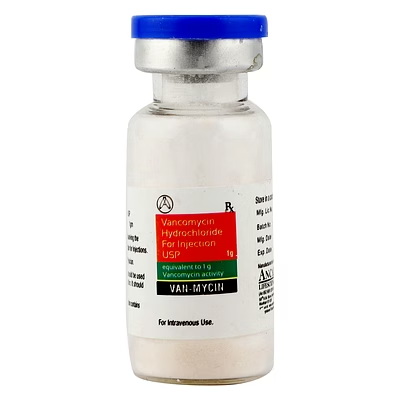
Vancomycin Hydrochloride for Injection USP 1gm
Vancomycin Hydrochloride for Injection USP
Vancomycin Hydrochloride Injection USP is a biocompatible parenteral antibiotic formulation designed for systemic bacterial infection management, delivering a precise 1gm dosage per vial for clinical reliability.
The standardized 1gm unit volume allows consistent therapeutic delivery, ensuring predictable pharmacodynamic outcomes in hospital and institutional applications.
Key Features:
- Optimized injectable for rapid systemic absorption
- 1gm active concentration per vial for standardized dosing
- Maintains chemical and physical stability under ambient and refrigerated storage
- Controlled osmolarity for minimal injection site irritation
Attributes:
- 1gm dosage ensuring reproducible therapeutic efficacy
- Injectable formulation with uniform solubility and flow characteristics
- Regulatory-compliant for bulk, hospital, and institutional use
Vancomycin Hydrochloride for Injection USP 500mg
Vancomycin Hydrochloride for Injection USP
Vancomycin Hydrochloride Injection USP is a biocompatible parenteral antibiotic formulation, delivering precise 500mg dosage per vial for systemic bacterial infection management.
The standardized 500mg volume allows repeatable dosing and predictable therapeutic performance in clinical applications.
Key Features:
- Optimized injectable for rapid systemic absorption
- 500mg active concentration per vial
- Maintains chemical and physical stability under storage
- Minimized injection discomfort with controlled osmolarity
Attributes:
- 500mg dosage for reproducible therapeutic efficacy
- Injectable formulation with controlled solubility
- Regulatory-compliant for hospital and institutional use
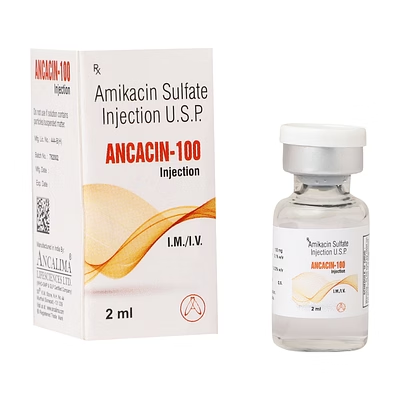
Amikacin Injection 100mg/2ml
Amikacin Injection
Amikacin Injection is a biocompatible injectable formulation designed for systemic antibacterial therapy, delivering precise 100mg/2ml dosage per injection.
The 2ml standardized volume provides repeatable dosing, ensuring predictable pharmacokinetic performance in clinical settings.
Key Features:
- Optimized aqueous formulation for rapid absorption
- 100mg active concentration per 2ml unit
- Maintains chemical and physical stability under ambient storage
- Minimized injection site discomfort with controlled osmolarity
Attributes:
- 100mg/2ml dosage for precise therapeutic administration
- Injectable formulation with consistent homogeneity
- Compliant with institutional and hospital pharmaceutical standards
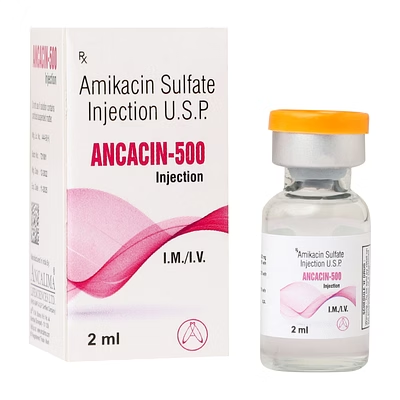
Amikacin Injection 500mg/2ml
Amikacin Injection
Amikacin Injection is a biocompatible parenteral aminoglycoside antibiotic formulated for systemic bacterial infections, delivering precise 500mg/2ml dosage per injection.
The 2ml standardized volume ensures accurate dosing, supporting reproducible clinical outcomes in professional medical environments.
Key Features:
- Optimized aqueous injectable for rapid systemic absorption
- 500mg active concentration per 2ml unit
- Maintains chemical and physical stability under standard storage
- Minimal injection site discomfort with optimized osmolarity
Attributes:
- 500mg/2ml for predictable therapeutic efficacy
- Injectable formulation with controlled viscosity
- Regulatory-compliant for institutional and hospital use
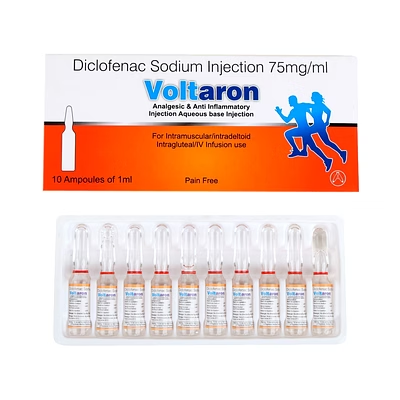
Diclofenac Sodium Injection 75mg/1ml
Diclofenac Sodium Injection
Diclofenac Sodium Injection is a biocompatible injectable formulation designed for rapid anti-inflammatory and analgesic therapy, delivering precise 75mg/1ml dosage per injection.
The 1ml standardized volume ensures consistent clinical effect while facilitating repeatable dosing in professional medical settings.
Key Features:
- Optimized aqueous formulation for rapid systemic absorption
- Consistent 75mg active concentration per milliliter
- Maintains physicochemical stability under standard storage
- Minimal injection site discomfort due to optimized pH
Attributes:
- 75mg/1ml concentration for predictable therapeutic efficacy
- Injectable formulation with controlled viscosity
- Regulated pharmaceutical-grade compliance
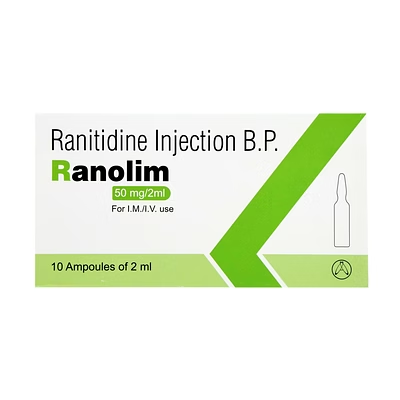
Ranitidine Injection 50mg/2ml
Ranitidine Injection
Ranitidine Injection is a biocompatible injectable formulation for targeted H2 receptor antagonism, facilitating gastric acid suppression in acute clinical applications.
The 50mg/2ml volume provides a standardized dose, enabling precise administration and predictable therapeutic efficacy.
Key Features:
- Optimized aqueous injectable for rapid absorption
- 50mg active concentration per 2ml unit
- Physicochemical stability ensures reproducible clinical effect
- Regulated for professional institutional use
Attributes:
- 50mg/2ml for accurate dosing
- Injectable formulation with controlled flow characteristics
- Compliance with pharmaceutical quality standards
Combipack of Ampicillin Injection 1gm with 10ml FFS WFI
Combipack of Ampicillin Injection
Combipack of Ampicillin Injection is a biocompatible injectable formulation designed for systemic antibacterial therapy, integrating Ampicillin in a sterile 10ml FFS WFI diluent for precision dosing.
The 1gm active dosage within the 10ml vehicle provides standardized therapeutic volume, ensuring accurate administration and consistent pharmacodynamic activity across clinical applications.
Key Features:
- Pre-measured ampoules for precise dosing
- Stable suspension in sterile Water for Injection
- Optimized for systemic antibacterial coverage
- Ambient and cold-chain storage stability
Attributes:
- 1gm active Ampicillin per 10ml FFS WFI
- FFS sterile formulation ensuring minimal contamination risk
- Institutional and clinical grade compliance
Injectable Solutions
Bulk Injection Products in the injectable solutions category include liquid formulations intended for parenteral administration. These products are sourced by hospitals, distributors, and OEM pharmaceutical companies requiring sterile production and controlled handling environments.
Formulation and production capabilities
Buyers evaluate injectable solution suppliers based on sterile processing capability, formulation stability, and production consistency. Considerations typically include manufacturing environment controls and documented procedures rather than finished-product branding or dosage customization.
Injectable Vaccines
Injectable Vaccines supplied in bulk form part of institutional and public health procurement channels. These products are sourced for regulated distribution programs, international supply contracts, and large-scale healthcare deployment initiatives.
Cold-chain and export readiness
Vaccine sourcing requires attention to cold-chain handling, export documentation, and logistics coordination. Buyers typically assess whether suppliers support temperature-controlled distribution and provide necessary compliance documentation for cross-border pharmaceutical movement.
Parenteral Drugs
Parenteral Drugs include injectable pharmaceuticals administered outside the gastrointestinal tract. This category supports hospital systems, emergency care providers, and specialized treatment facilities sourcing injectable medications in bulk quantities.
Regulatory compliance and safety
Bulk parenteral drug procurement focuses on sterility assurance, regulatory documentation, and safety controls. Buyers generally review compliance alignment with importing market regulations and internal quality systems before finalizing sourcing decisions.
Sterile Injections
Sterile Injections encompass injectable products manufactured under strict aseptic conditions. These are commonly sourced by distributors and institutional buyers managing high-volume or multi-location healthcare supply chains.
Bulk supply and packaging standards
Bulk sterile injection sourcing emphasizes packaging integrity, handling standards, and batch traceability. Buyers assess packaging formats, shipment suitability, and supply consistency to ensure alignment with their storage and distribution infrastructure.