Combipack of Artesunate for Injection 60mg
Artesunate Injection
Artesunate Injection is a biocompatible antimalarial injectable formulation delivering 60mg per pack, designed for rapid parasiticidal action in clinical and hospital environments.
The 60mg specification ensures precise therapeutic dosing for institutional and emergency medical use.
Key Features:
- Rapid systemic antimalarial effect
- Stable under ambient and refrigerated storage
- Low-viscosity solution for smooth administration
Attributes:
- 60mg per pack for predictable therapeutic outcome
- Injectable solution with uniform solubility
- Compliance-ready for bulk hospital and institutional procurement
Betaderm MN Cream 25gm
Key Features of Betaderm MN Cream 25gm
- Topical cream formulation
- Pack Size: 25gm
- Suitable for bulk and wholesale orders
Product Attributes & Specifications for Bulk Supply
- Format: Topical Cream
- Intended Users: Distributors, OEMs, Exporters
Combipack of Ceftriaxone 1gm with 10ml FFS WFI
Ondansetron Injection 8mg/4ml
Ondansetron Injection
Ondansetron Injection is a biocompatible parenteral formulation designed for antiemetic therapy, providing precise 8mg/4ml dosing for rapid onset of action in clinical settings.
The 4ml standardized volume ensures repeatable dosing and consistent therapeutic effect.
Key Features:
- Optimized aqueous injectable for rapid systemic absorption
- 8mg active concentration per 4ml unit
- Maintains stability under ambient and refrigerated storage
- Minimized injection site irritation via controlled pH
Attributes:
- 8mg/4ml for predictable therapeutic efficacy
- Injectable formulation with controlled viscosity
- Regulatory-compliant for professional institutional use
Miconazole Nitrate 2% Cream 30gm
Key Features of Miconazole Nitrate 2% Cream 30gm
- 2% miconazole nitrate concentration
- Topical cream formulation
- Pack Size: 30gm
- Bulk and wholesale supply ready
Product Attributes & Specifications for Bulk Procurement
- Primary Ingredient: Miconazole Nitrate
- Format: Topical Cream
- Intended Users: Distributors, OEMs, Exporters
Ketoconazole 2% Cream 30gm
Key Features of Ketoconazole 2% Cream 30gm
- 2% ketoconazole concentration
- Topical cream formulation
- Pack Size: 30gm
- Bulk and wholesale supply ready
Product Attributes & Specifications for Bulk Sourcing
- Primary Ingredient: Ketoconazole
- Format: Topical Cream
- Intended Users: Distributors, OEMs, Exporters
Paraldehyde Injection B.P. 5ml
Paraldehyde Injection B.P.
Paraldehyde Injection B.P. is a biocompatible injectable formulation for rapid central nervous system depressant therapy, delivering 5ml dosage per vial for controlled clinical use.
The 5ml specification ensures precise administration and predictable therapeutic effect for emergency and institutional applications.
Key Features:
- Rapid CNS depressant action with controlled dosing
- Maintains chemical and physical stability under ambient storage
- Standardized solution for reliable therapeutic outcomes
Attributes:
- 5ml per vial for precise dosing
- Injectable formulation with uniform flow characteristics
- Compliance-ready for bulk, hospital, and institutional use
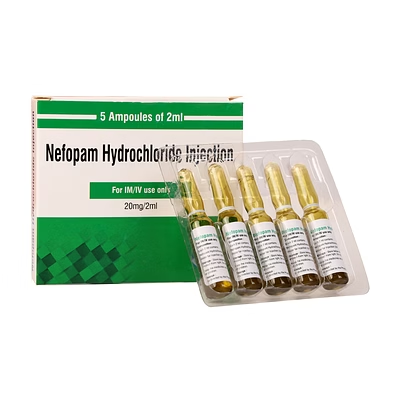
Nefopam Hydrochloride Injection 20mg/2ml
Nefopam Hydrochloride Injection
Nefopam Hydrochloride Injection is a biocompatible parenteral analgesic formulation delivering 20mg per 2ml for rapid pain management in clinical and hospital settings.
The 20mg/2ml specification ensures precise dosing and predictable therapeutic outcomes in institutional applications.
Key Features:
- Rapid systemic analgesic action with controlled dosing
- Stable injectable formulation under ambient and refrigerated storage
- Minimal tissue irritation due to controlled osmolarity
Attributes:
- 20mg per 2ml vial for consistent analgesic effect
- Injectable solution with uniform flow and solubility
- Compliance-ready for bulk, hospital, and institutional use
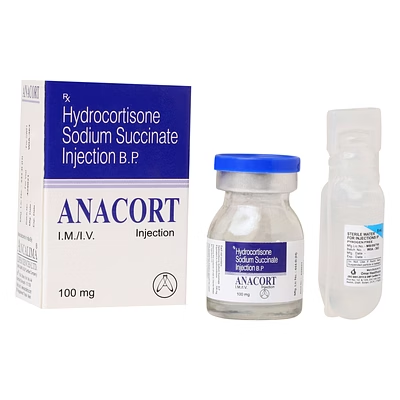
Hydrocortisone Sodium Succinate Injection 100mg
Hydrocortisone Sodium Succinate Injection
Hydrocortisone Sodium Succinate Injection is a biocompatible injectable formulation designed for rapid systemic corticosteroid delivery, providing precise 100mg dosing per vial for clinical applications.
The 100mg specification ensures standardized anti-inflammatory and immunosuppressive therapy, allowing predictable pharmacokinetics in hospital and institutional use.
Key Features:
- Optimized solubility for immediate systemic absorption
- Maintains chemical and physical stability under storage conditions
- Controlled pH and osmolarity for minimal injection site irritation
- Single-use vial for dosage accuracy and safety
Attributes:
- 100mg dosage ensuring consistent therapeutic effect
- Injectable formulation with uniform flow and stability
- Compliance-ready for bulk, hospital, and institutional use
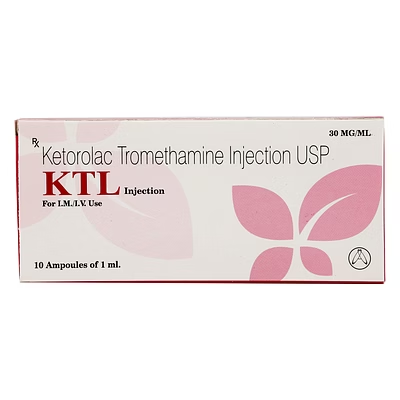
Ketorolac Tromethamine Injection 30mg/1ml
Ketorolac Tromethamine Injection
Ketorolac Tromethamine Injection is a biocompatible injectable NSAID formulation delivering 30mg per 1ml vial, designed for rapid analgesic and anti-inflammatory effect in controlled clinical environments.
The 30mg/1ml specification ensures standardized dosing for predictable pain management in hospital and institutional use.
Key Features:
- Rapid onset analgesic effect with standardized dosing
- Stable chemical composition under ambient and refrigerated conditions
- Controlled pH and osmolarity for injection safety
Attributes:
- 30mg per 1ml vial for predictable therapeutic outcomes
- Injectable formulation with uniform solubility and flow
- Suitable for bulk hospital and institutional procurement
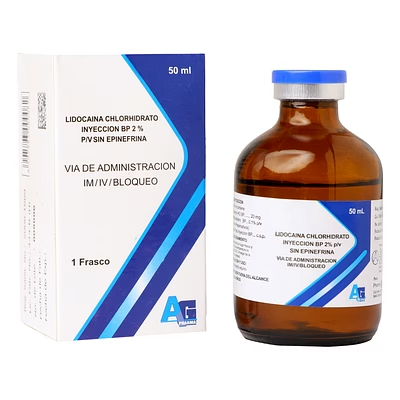
Lidocaine Injection BP 2% w/v
Lidocaine Injection BP
Lidocaine Injection BP is a biocompatible injectable local anesthetic delivering 2% w/v solution, designed for rapid onset analgesia and procedural anesthesia in clinical environments.
The 2% w/v specification ensures standardized anesthetic effect, suitable for surgical, diagnostic, and institutional applications.
Key Features:
- Rapid onset of local anesthesia
- Stable formulation under ambient and refrigerated storage
- Optimized pH and osmolarity for minimal tissue irritation
Attributes:
- 2% w/v concentration for predictable anesthetic effect
- Injectable solution with uniform flow and solubility
- Compliance-ready for bulk hospital and institutional use
Combipack of Artesunate for Injection 120mg
Artesunate Injection
Artesunate Injection is a biocompatible antimalarial injectable formulation delivering 120mg per pack, designed for rapid parasiticidal action in clinical and hospital settings.
The 120mg specification ensures precise therapeutic dosing, suitable for institutional and emergency use.
Key Features:
- Rapid systemic antimalarial activity
- Stable formulation under ambient and refrigerated conditions
- Low-viscosity solution for smooth injection
Attributes:
- 120mg per pack for predictable therapeutic effect
- Injectable formulation with consistent solubility
- Compliance-ready for bulk hospital and institutional procurement
Triamcinolone Acetonide Injectable Suspension USP 40mg/1ml
Triamcinolone Acetonide Injectable Suspension USP
Triamcinolone Acetonide Injectable Suspension USP is a biocompatible pharmaceutical formulation engineered for targeted corticosteroid delivery via intramuscular or intra-articular injection.
The 40mg/1ml concentration provides a standardized clinical dosage, enabling precise administration while minimizing dosing variability and ensuring consistent therapeutic efficacy.
Key Features:
- Optimized suspension for uniform corticosteroid dispersion
- Controlled particle size for predictable pharmacokinetics
- Viscosity engineered to minimize injection resistance
- Stable under ambient and refrigerated storage conditions
Attributes:
- 40mg/1ml dosage for precise clinical application
- Injectable suspension with consistent homogeneity
- Regulated pharmaceutical compliance for institutional use
Tranexamic Acid Injection BP (100mg/ml)
Tranexamic Acid Injection BP
Tranexamic Acid Injection BP is a biocompatible injectable formulation designed to inhibit fibrinolysis and stabilize clot formation. The 100mg/ml concentration ensures precise dosing for clinical hemostatic interventions.
This standardized dosage supports rapid hemostatic control, making it suitable for surgical, trauma, and institutional applications.
Key Features:
- High solubility and stability in isotonic solution
- Optimized for rapid systemic fibrinolytic inhibition
- Minimizes injection site irritation due to controlled pH
- Single-use vial ensures dosage accuracy
Attributes:
- 100mg/ml per vial for predictable therapeutic effect
- Injectable formulation with uniform flow
- Suitable for bulk hospital and institutional procurement
Magnesium Sulphate Injection BP 50% w/v 5ml
Magnesium Sulphate Injection BP
Magnesium Sulphate Injection BP is a biocompatible injectable electrolyte solution delivering 50% w/v in 5ml vials, designed for rapid correction of magnesium deficiency and seizure prophylaxis.
The 50% w/v, 5ml specification allows for precise therapeutic dosing in institutional and hospital settings.
Key Features:
- High-purity magnesium sulphate for rapid systemic effect
- Optimized isotonicity and pH for injection safety
- Stable under ambient and refrigerated conditions
Attributes:
- 50% w/v in 5ml per vial for accurate dosing
- Low-viscosity injectable solution for smooth administration
- Bulk-sourcing ready for hospitals and institutional procurement
Combipack of Pantoprazole for Injection 40mg
Combipack of Pantoprazole for Injection 40mg - Injectable Formulation
Pantoprazole injectable combipack contains 40mg per unit in sterile injectable form. Suitable for professional clinical use and bulk procurement.
Supports distributors, OEMs, and export buyers requiring standardized injectable supply with secure handling.
Key Features
- Sterile injectable combipack
- 40mg per unit
- Bulk and institutional supply ready
Product Attributes & Specifications
- Formulation: Sterile injectable solution
- Composition: Pantoprazole
- Packaging: Combipack format
Magnesium Sulphate Injection BP 50% w/v 10ml
Magnesium Sulphate Injection BP 50% w/v 10ml - Injectable Formulation
Magnesium Sulphate Injection BP provides a sterile 50% w/v solution in 10ml vials. Designed for professional clinical and bulk procurement use.
Suitable for distributors, institutional buyers, OEMs, and export supply requiring standardized injectable formats.
Key Features
- Sterile 50% w/v injectable solution
- 10ml vial per unit
- Bulk and institutional supply ready
Product Attributes & Specifications
- Composition: Magnesium Sulphate BP
- Formulation: Injectable solution
- Packaging: 10ml vial
Gentamicin Injection B.P. 2ml (80mg/2ml)
Gentamicin Injection B.P. 2ml (80mg/2ml) - Injectable Formulation
Gentamicin Injection B.P. provides 80mg per 2ml vial in a sterile injectable formulation. Designed for professional clinical settings and bulk procurement.
Suitable for distributors, institutional buyers, OEMs, and export supply chains requiring standardized handling and professional-grade injectable products.
Key Features
- Sterile 2ml injectable solution
- Contains 80mg Gentamicin per vial
- Prepared for bulk and institutional supply
Product Attributes & Specifications
- Formulation: Injectable solution
- Intended for clinical use
- Packaging: single 2ml vial
Dexamethasone Injection USP (4mg/1ml) 1ml
Dexamethasone Injection USP (4mg/1ml) 1ml - Injectable Formulation
Dexamethasone Injection USP provides 4mg per 1ml vial in sterile injectable form. Prepared for professional clinical and bulk procurement usage.
Supports distributors, institutional buyers, OEMs, and export buyers needing standardized handling and professional injectable formats.
Key Features
- Sterile 1ml injectable solution
- 4mg per unit dosage
- Bulk and institutional supply ready
Product Attributes & Specifications
- Formulation: USP sterile injectable solution
- Composition: Dexamethasone
- Packaging: 1ml vial
Combipack of Omeprazole Sodium for Injection 40mg
Combipack of Omeprazole Sodium for Injection 40mg - Injectable Formulation
This combipack provides sterile 40mg Omeprazole Sodium per unit in injectable form. Prepared for bulk procurement and professional clinical usage.
Designed to support distributors, OEMs, and export buyers requiring efficient handling and standardized packaging for institutional supply.
Key Features
- Sterile injectable combipack
- 40mg per unit dosage
- Bulk and institutional-ready format
Product Attributes & Specifications
- Formulation: Injectable solution
- Composition: Omeprazole Sodium
- Packaging: Combipack format
Vancomycin Hydrochloride for Injection USP 1gm
Vancomycin Hydrochloride for Injection USP
Vancomycin Hydrochloride Injection USP is a biocompatible parenteral antibiotic formulation designed for systemic bacterial infection management, delivering a precise 1gm dosage per vial for clinical reliability.
The standardized 1gm unit volume allows consistent therapeutic delivery, ensuring predictable pharmacodynamic outcomes in hospital and institutional applications.
Key Features:
- Optimized injectable for rapid systemic absorption
- 1gm active concentration per vial for standardized dosing
- Maintains chemical and physical stability under ambient and refrigerated storage
- Controlled osmolarity for minimal injection site irritation
Attributes:
- 1gm dosage ensuring reproducible therapeutic efficacy
- Injectable formulation with uniform solubility and flow characteristics
- Regulatory-compliant for bulk, hospital, and institutional use
Vancomycin Hydrochloride for Injection USP 500mg
Vancomycin Hydrochloride for Injection USP
Vancomycin Hydrochloride Injection USP is a biocompatible parenteral antibiotic formulation, delivering precise 500mg dosage per vial for systemic bacterial infection management.
The standardized 500mg volume allows repeatable dosing and predictable therapeutic performance in clinical applications.
Key Features:
- Optimized injectable for rapid systemic absorption
- 500mg active concentration per vial
- Maintains chemical and physical stability under storage
- Minimized injection discomfort with controlled osmolarity
Attributes:
- 500mg dosage for reproducible therapeutic efficacy
- Injectable formulation with controlled solubility
- Regulatory-compliant for hospital and institutional use
Amikacin Injection 100mg/2ml
Amikacin Injection
Amikacin Injection is a biocompatible injectable formulation designed for systemic antibacterial therapy, delivering precise 100mg/2ml dosage per injection.
The 2ml standardized volume provides repeatable dosing, ensuring predictable pharmacokinetic performance in clinical settings.
Key Features:
- Optimized aqueous formulation for rapid absorption
- 100mg active concentration per 2ml unit
- Maintains chemical and physical stability under ambient storage
- Minimized injection site discomfort with controlled osmolarity
Attributes:
- 100mg/2ml dosage for precise therapeutic administration
- Injectable formulation with consistent homogeneity
- Compliant with institutional and hospital pharmaceutical standards
Amikacin Injection 500mg/2ml
Amikacin Injection
Amikacin Injection is a biocompatible parenteral aminoglycoside antibiotic formulated for systemic bacterial infections, delivering precise 500mg/2ml dosage per injection.
The 2ml standardized volume ensures accurate dosing, supporting reproducible clinical outcomes in professional medical environments.
Key Features:
- Optimized aqueous injectable for rapid systemic absorption
- 500mg active concentration per 2ml unit
- Maintains chemical and physical stability under standard storage
- Minimal injection site discomfort with optimized osmolarity
Attributes:
- 500mg/2ml for predictable therapeutic efficacy
- Injectable formulation with controlled viscosity
- Regulatory-compliant for institutional and hospital use
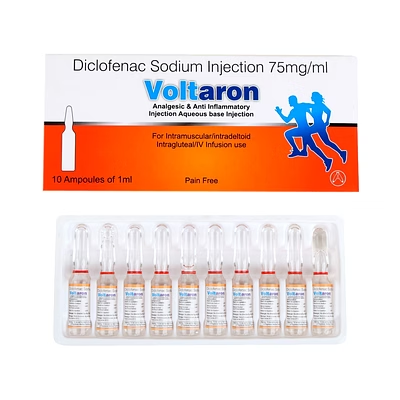
Diclofenac Sodium Injection 75mg/1ml
Diclofenac Sodium Injection
Diclofenac Sodium Injection is a biocompatible injectable formulation designed for rapid anti-inflammatory and analgesic therapy, delivering precise 75mg/1ml dosage per injection.
The 1ml standardized volume ensures consistent clinical effect while facilitating repeatable dosing in professional medical settings.
Key Features:
- Optimized aqueous formulation for rapid systemic absorption
- Consistent 75mg active concentration per milliliter
- Maintains physicochemical stability under standard storage
- Minimal injection site discomfort due to optimized pH
Attributes:
- 75mg/1ml concentration for predictable therapeutic efficacy
- Injectable formulation with controlled viscosity
- Regulated pharmaceutical-grade compliance
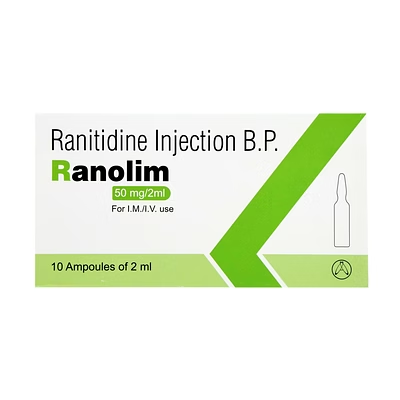
Ranitidine Injection 50mg/2ml
Ranitidine Injection
Ranitidine Injection is a biocompatible injectable formulation for targeted H2 receptor antagonism, facilitating gastric acid suppression in acute clinical applications.
The 50mg/2ml volume provides a standardized dose, enabling precise administration and predictable therapeutic efficacy.
Key Features:
- Optimized aqueous injectable for rapid absorption
- 50mg active concentration per 2ml unit
- Physicochemical stability ensures reproducible clinical effect
- Regulated for professional institutional use
Attributes:
- 50mg/2ml for accurate dosing
- Injectable formulation with controlled flow characteristics
- Compliance with pharmaceutical quality standards
Combipack of Ampicillin Injection 1gm with 10ml FFS WFI
Combipack of Ampicillin Injection
Combipack of Ampicillin Injection is a biocompatible injectable formulation designed for systemic antibacterial therapy, integrating Ampicillin in a sterile 10ml FFS WFI diluent for precision dosing.
The 1gm active dosage within the 10ml vehicle provides standardized therapeutic volume, ensuring accurate administration and consistent pharmacodynamic activity across clinical applications.
Key Features:
- Pre-measured ampoules for precise dosing
- Stable suspension in sterile Water for Injection
- Optimized for systemic antibacterial coverage
- Ambient and cold-chain storage stability
Attributes:
- 1gm active Ampicillin per 10ml FFS WFI
- FFS sterile formulation ensuring minimal contamination risk
- Institutional and clinical grade compliance
ELMOLIM Moisturizing Cream 100 gm
ELMOLIM Moisturizing Cream
ELMOLIM Moisturizing Cream is a topical formulation engineered for uniform hydration across larger skin areas. The 100gm specification provides sufficient volume for repeated clinical or professional use.
The cream ensures consistent spreadability and stability during bulk handling.
Key Features
- Uniform hydration delivery
- Stable formulation for extended use
- Optimized for bulk and OEM sourcing
Attributes
- Formulation Type: Topical Cream
- Quantity: 100gm
- Order Capability: Bulk/Wholesale, OEM compatible
INCRETON Beauty Cream 60 gm
INCRETON Beauty Cream
INCRETON Beauty Cream is a topical formulation designed for precision application across facial and body areas. The 60gm specification allows repeated professional usage while maintaining consistent performance.
The formulation ensures smooth texture, spreadability, and stability.
Key Features
- Consistent topical coverage
- Stable formulation for multiple applications
- Optimized for bulk and OEM orders
Attributes
- Formulation Type: Topical Cream
- Quantity: 60gm
- Order Capability: Bulk/Wholesale, OEM compatible
ELIRASH Anti-Rash Cream 40gm
ELIRASH Anti-Rash Cream
ELIRASH Anti-Rash Cream is a topical formulation providing targeted relief and skin protection. The 40gm specification allows precise localized application for institutional or clinical use.
The formulation ensures consistent absorption, spreadability, and long-term stability.
Key Features
- Targeted topical delivery
- Stable formulation for repeated use
- Optimized for bulk procurement
Attributes
- Formulation Type: Topical Cream
- Quantity: 40gm
- Order Capability: Bulk/Wholesale, OEM compatible
DOLAVER Intimate Wash 100ml
DOLAVER Intimate Wash
DOLAVER Intimate Wash is a topical formulation optimized for delicate skin cleansing. The 100ml specification provides sufficient volume for repeated professional or institutional use, ensuring standardized application.
The formulation is engineered for consistent viscosity and skin-safe pH balance.
Key Features
- Standardized clinical volume
- Maintains skin pH balance
- Optimized for bulk institutional use
Attributes
- Formulation Type: Topical Wash
- Quantity: 100ml
- Order Capability: Bulk/Wholesale, OEM compatible
REJO-E Glowing Serum 30ml
REJO-E Glowing Serum
REJO-E Glowing Serum is a topical formulation designed for precision application and optimized skin absorption. The 30ml format provides consistent dosing for professional or clinical use.
The formulation ensures uniform spreadability and stability across repeated applications.
Key Features
- Optimized for controlled topical application
- Consistent serum absorption
- Maintains performance over multiple uses
Attributes
- Formulation Type: Topical Serum
- Quantity: 30ml
- Order Capability: Bulk/Wholesale, OEM compatible
SELOMA Under Eye Cream 15gm
SELOMA Under Eye Cream
SELOMA Under Eye Cream is a topical formulation designed for precise localized delivery around delicate periocular regions. The 15gm specification ensures controlled application and minimizes wastage.
The formulation provides consistent spreadability, absorption, and hydration performance tailored to professional cosmetic applications.
Key Features
- Localized precision delivery
- Uniform topical consistency
- Optimized for repeat clinical or salon use
Attributes
- Formulation Type: Topical Cream
- Quantity: 15gm
- Order Capability: Bulk/Wholesale, OEM compatible
Active Pharmaceutical Ingredients
Bulk Pharmaceuticals in this category include Active Pharmaceutical Ingredients used as base compounds in finished drug manufacturing. These materials are typically sourced by pharmaceutical manufacturers, formulators, and contract production units supplying regulated and semi-regulated markets. The category focuses on bulk pharmaceutical sourcing rather than finished dosage branding or retail packaging.
Manufacturing capabilities and certifications
API sourcing in this category is aligned with bulk production environments where consistency, batch traceability, and documented manufacturing processes are required. Buyers typically assess suppliers based on process control standards, documentation availability, and alignment with destination-market regulatory expectations without assuming product-specific approvals.
Generic Medicines
Generic Medicines under Bulk Pharmaceuticals refer to non-branded pharmaceutical formulations supplied for institutional procurement, redistribution, or secondary manufacturing. These products are commonly sourced for public health supply chains, hospital networks, and international distributors focusing on cost-effective therapeutic availability.
Use cases and applications
Bulk generic medicines are often procured for large-scale distribution, government tenders, or OEM pharmaceutical manufacturing. Buyers typically evaluate suitability based on formulation category, dosage form grouping, and regulatory compatibility with their target markets rather than individual product performance attributes.
OTC Pharmaceuticals
OTC Pharmaceuticals in this category include bulk-supplied formulations intended for over-the-counter distribution channels. These products are positioned for markets where non-prescription access is permitted and are generally sourced by wholesalers, private label marketers, and international pharmaceutical distributors.
Export readiness and compliance
OTC pharmaceutical sourcing at a bulk level requires attention to labeling adaptability, documentation readiness, and compliance alignment for importing regions. Buyers typically confirm export documentation processes and regulatory references applicable to their destination markets before finalizing supply arrangements.
Pharmaceutical Excipients
Pharmaceutical Excipients cover non-active substances used in formulation stability, delivery, and processing. This category supports manufacturers producing tablets, capsules, injectables, and topical products where excipient quality directly affects manufacturing consistency rather than therapeutic outcomes.
Quality and sourcing standards
Bulk excipient sourcing focuses on material consistency, contamination control, and documented sourcing practices. Buyers commonly assess excipients based on formulation compatibility, supply continuity, and adherence to recognized pharmacopeial or internal quality benchmarks relevant to their manufacturing processes.